
September 8, 2025
Navigating Malaysia’s pharmaceutical and healthcare market demands more than just a quality product—it requires strategic compliance with the country’s regulatory framework. The National Pharmaceutical Regulatory Agency (NPRA) stands as the gatekeeper, ensuring that every medicine, supplement, or skincare product meets strict safety, efficacy, and quality benchmarks before reaching consumers.
For businesses—whether a pharmacy distributor in Malaysia, an international brand, or a local manufacturer—this process isn’t just bureaucratic red tape. It’s the proven foundation for securing market access, building trusted partnerships, and ensuring long-term viability in a rapidly growing sector.
Why does this matter?
Malaysia’s healthcare market is expanding, driven by:
Rising consumer awareness of preventive healthcare
Increased demand for specialized supplements and skincare
Strengthened pharmacy distribution networks
Consider this real-world scenario: A local distributor importing a new line of collagen supplements from Japan faces delays of 6–12 months due to incomplete NPRA documentation. The financial impact? Missed revenue opportunities, strained retailer relationships, and potential brand credibility loss.
Key Statistics:
Malaysia’s pharmaceutical market is projected to grow at 6.3% CAGR (2023–2030).
Over 70% of rejected applications stem from misclassification or incomplete dossiers.
For pharmacy wholesale distributors, this means one thing: Efficient registration isn’t optional—it’s a competitive edge.

The NPRA operates under Malaysia’s Ministry of Health (MOH), enforcing regulations through its Drug Registration Guidance Document (DRGD)—a comprehensive blueprint for product approvals.
What does the DRGD cover?
Registration pathways (new drugs, generics, supplements)
Technical requirements (dossier structure, stability data)
Post-market obligations (renewals, adverse event reporting)
For pharmacy distribution services in Malaysia, mastering the DRGD is non-negotiable. A single oversight—like incorrect stability testing protocols—can derail an entire submission.
The Registration Journey (Step-by-Step):
Product Categorization – Is it a drug, supplement, or cosmetic?
Dossier Preparation – Compiling Modules 1–5 in Common Technical Document (CTD) format.
QUEST3+ Submission – NPRA’s online portal for application tracking.
Scientific Evaluation – Review of quality, safety, and efficacy data.
Local Example: A Kuala Lumpur-based importer of German probiotics reduced approval time by 40% by pre-validating their GMP certificates and labeling artwork before submission.
Why This Works:
Reliable adherence to NPRA’s framework minimizes risks.
Strategic dossier preparation accelerates market entry.
Learn More : Global Benchmarking Tool (GBT) for Evaluation of National Regulatory Systems
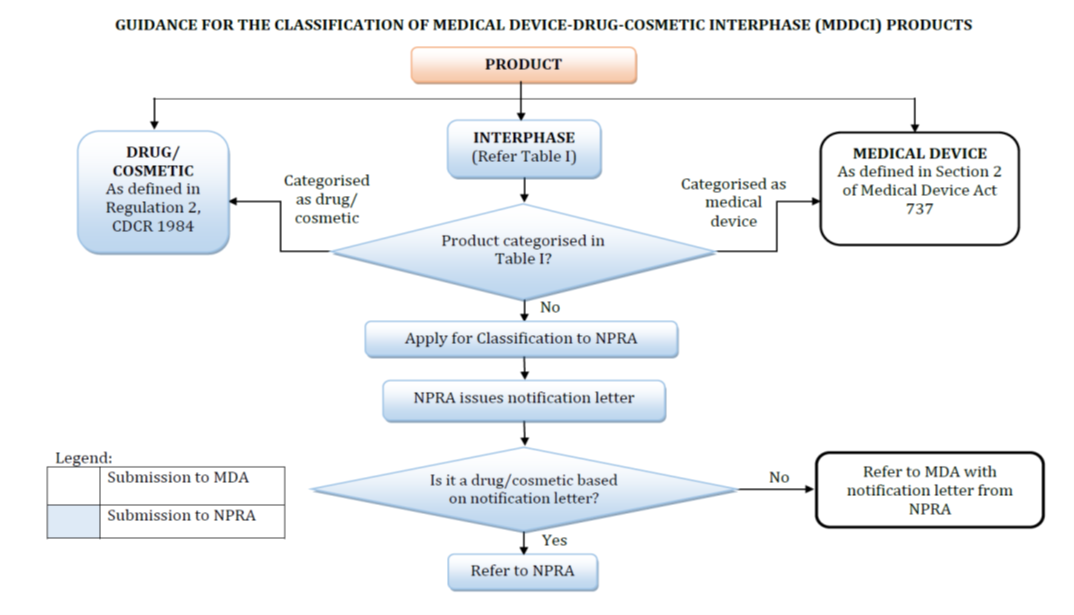
Misclassification is the #1 reason for NPRA rejections. A health supplement with implied therapeutic claims could be reclassified as a drug, triggering costly reformulation or testing.
NPRA’s Product Categories:
Prescription Drugs (e.g., antibiotics, specialized medications)
Over-the-Counter (OTC) Medicines (e.g., pain relievers, antacids)
Health Supplements (e.g., vitamins, probiotics)
Traditional Medicines (e.g., herbal remedies)
Natural Products (e.g., plant-based extracts)
Common Pitfalls:
A skincare serum marketed with “anti-inflammatory” claims may require drug registration.
Energy drinks with high caffeine levels could face supplement regulations.
Operational Scenario:
A Malaysian distributor of Thai herbal supplements initially applied under “Traditional Medicine” but was redirected to “Health Supplements” after NPRA flagged unapproved efficacy claims. The delay cost 3 months in lost sales.
Pro Tip:
Use NPRA’s Medicinal Product Classification Committee (MPCC) or the QUEST portal for pre-submission clarity.
Learn More : KKM Approval Process for Skincare and Supplements | Medicine Classification Guidelines

A flawless dossier is your ticket to approval. The Common Technical Document (CTD) format is mandatory, split into:
Module 1: Administrative Documents
Application forms
Proof of licensing (manufacturer, importer)
Local agent appointment letter (for foreign brands)
Modules 2–5: Scientific Data
Quality (GMP certificates, batch testing)
Safety (toxicology reports, side-effect profiles)
Efficacy (clinical trials, literature reviews)
Critical Documents Checklist:
✅ GMP Certificates (from approved facilities)
✅ Free Sale Certificate (from the country of origin)
✅ Product Formula (full ingredient disclosure)
✅ Stability Studies (shelf-life validation)
✅ Labeling Artwork (compliant with Malaysian guidelines)
Case Study:
A Selangor-based pharmacy distributor faced rejection due to untranslated Japanese stability reports. After re-submitting with certified translations, approval was granted in 4 months.
Why Detail Matters:
80% of delays stem from incomplete or unverified data.
Expert preparation reduces back-and-forth queries.
Learn More : Guidance on Use of PRISM for Regulatory Submission
Who needs what license?
Manufacturer’s License – For local production.
Import License – For overseas brands.
Wholesaler’s License – For third-party distribution.
Foreign Brands: Must appoint a NPRA-registered local agent to:
Submit applications
Handle post-market surveillance
Manage renewals
Penang-Based Example:
A Swiss vitamin brand partnered with a Klang Valley distributor to secure their import license and local agent representation, cutting approval time by 30%.
Key Takeaways:
Efficient licensing aligns with long-term compliance.
Tailored partnerships (e.g., local agents) streamline operations.
Navigating Malaysia’s digital submission system for pharmaceutical registrations can feel like decoding a complex puzzle—especially for first-time applicants. The QUEST3+ platform, used by the National Pharmaceutical Regulatory Agency (NPRA), is the proven gateway for all product registrations. While it’s an efficient system, its technical nuances can trip up even experienced pharmacy distributors.
Here’s how to master the process:
Company Registration – Before anything else, businesses must register with NPRA and obtain login credentials. This step is non-negotiable—without it, you can’t even start.
Product Information Entry – Every detail, from ingredient lists to manufacturing sites, must be accurately input. A single typo can trigger delays.
Document Uploads – The Common Technical Document (CTD) format is mandatory. Missing even one module means automatic rejection.
Fee Payment – NPRA charges a processing fee, payable via FPX or bank transfer. Late payments pause your application.
Submission & Tracking – Once submitted, the QUEST3+ dashboard becomes your lifeline for updates.
Real-World Challenge:
A Johor Bahru-based distributor of Australian vitamins faced three rejections due to incorrect file formats in their stability reports. After switching to PDF/A-compliant files, approval came in 8 months.
Why This Matters:
80% of technical rejections stem from upload errors.
Strategic preparation cuts processing time by 30–50%.
Pro Tip:
Assign an internal compliance officer to pre-audit submissions before hitting “submit.”
Learn More : Top 10 Pharmaceutical Companies in Malaysia 2024 | Pharmacy Retail Merchandising Servicers: Top 10 Picks for 2025

Hitting “submit” is just the first milestone. NPRA’s evaluation phase is where most applications stall—unless you’ve preempted every question.
The 3-Pillar Assessment:
Quality – Is the product manufactured under GMP? Are batch records consistent?
Safety – Does the toxicology data justify the dosage? Are side effects properly disclosed?
Efficacy – For drugs, clinical trials are critical. For supplements, literature reviews may suffice.
Upon Approval:
A 5-year registration certificate is issued.
Labels and packaging must display the NPRA registration number.
Post-market surveillance begins immediately.
Local Example:
A Kuala Lumpur skincare brand reformulated their acne treatment but forgot NPRA’s variation approval process. The oversight led to a voluntary recall and 6-month sales freeze.
Key Takeaway:
Post-approval compliance is just as critical as the initial submission.
Expert oversight prevents costly mistakes.

Why do 40% of applications face delays or rejections? The answer often lies in avoidable missteps.
Top 4 Roadblocks:
Incomplete Dossiers – Missing GMP certificates or free sale documents.
Misclassification – Calling a supplement a “drug” (or vice versa).
Slow Response Times – NPRA queries often get buried in inboxes.
Import License Gaps – Foreign brands underestimating local agent requirements.
Solutions That Work:
✅ Pre-Submission Audits – A third-party review catches 90% of errors.
✅ Dedicated Regulatory Teams – One person should own NPRA communications.
✅ Local Agent Partnerships – They navigate bureaucracy on your behalf.
Case Study:
A Penang-based pharmacy distributor cut their approval timeline from 18 to 9 months by hiring a regulatory consultant to pre-screen their dossier.
Power Move:
Strategic partnerships turn hurdles into routine checkpoints.
Registration isn’t the finish line—it’s the first lap. Pharmacy wholesale distributors play a pivotal role in maintaining compliance long after approval.
Ongoing Duties:
Batch Monitoring – Tracking recalls and adverse reactions.
Renewal Management – Submitting updated stability data before the 5-year expiry.
Documentation Storage – NPRA can audit records anytime.
Operational Reality:
Smaller independent pharmacy distributors often lack in-house expertise, risking non-compliance. The solution? Outsourcing to trusted compliance firms.
Why It Pays Off:
Regulatory goodwill speeds up future approvals.
Consumer trust grows when safety protocols are visible.
Learn More : Securing Pharma Supply Chains
In Malaysia’s crowded pharmaceutical landscape, compliance isn’t just a rule—it’s a differentiator. Brands that master NPRA’s framework gain:
Faster market entry than competitors.
Stronger retailer relationships (who prefer fully compliant partners).
Long-term consumer loyalty.
The Bottom Line:
Registration is just step one.
Ongoing vigilance keeps your brand in the clear.
Q1: What products require a KKM check?
Answer: Products such as cosmetics, health supplements, traditional medicines, over-the-counter (OTC) medicines, and medical devices require a KKM check to ensure they are safe, properly labeled, and registered with the Ministry of Health Malaysia.
Q2: How to check if a product is registered?
Answer: You can verify a product’s registration through the NPRA’s official database (QUEST system) by entering the product name, registration number, or company name.
Q3: How to register FDA in Malaysia?
Answer: Malaysia does not use the term “FDA.” Instead, product registration is handled by NPRA (for drugs, cosmetics, health supplements) and MDA (for medical devices). Companies must submit applications through the respective online systems (QUEST for NPRA, Medcast for MDA).
Q4: How to register NPRA in Malaysia?
Answer: To register with NPRA, companies must apply through the QUEST online portal, submit required documents (quality, safety, efficacy data), pay the registration fees, and await evaluation and approval.
Q5: What is a KKM-approved product?
Answer: A KKM-approved product is one that has been officially evaluated and registered by the Ministry of Health Malaysia, meeting safety, quality, and regulatory standards set by NPRA or MDA.
Q6: Which products need FDA approval?
Answer: In Malaysia, the equivalent authority is NPRA or MDA, not FDA. In the U.S., FDA approval is required for drugs, biologics, medical devices, food additives, and certain cosmetics.
Q7: What is Moh certified?
Answer: “MOH certified” refers to products or facilities that meet standards set by the Ministry of Health Malaysia, typically meaning they are approved or registered under the NPRA or MDA regulatory system.
Q8: What is a KKM certificate?
Answer: A KKM certificate is the official proof of registration issued by the Ministry of Health Malaysia, confirming that a product complies with regulatory requirements and is safe for consumer use.
Q9: What is MDA approval?
Answer: MDA approval is certification from the Medical Device Authority confirming that a medical device meets safety, performance, and regulatory standards for sale and use in Malaysia.
Q10: What is NPRA KKM?
Answer: NPRA (National Pharmaceutical Regulatory Agency) is a division under the Ministry of Health (KKM) responsible for regulating medicines, cosmetics, traditional products, and health supplements in Malaysia.
Ready to Simplify Your NPRA Journey?
At PriooCare Malaysia, we specialize in end-to-end regulatory support—from dossier preparation to post-market compliance. Whether you’re a global pharma brand or a local supplement startup, our tailored solutions ensure your products meet NPRA standards without the headaches. Contact us today to discuss how we can streamline your registration and distribution strategy in Malaysia.