
August 5, 2025
Creating a dependable pharmaceutical supply chain within Malaysia demands far more than simply offering quality products and ensuring timely deliveries. The absolute bedrock, the non-negotiable element, involves gaining a profound grasp of the intricate regulatory environment and rigorously adhering to its demands. Success hinges on this compliance foundation.
Malaysia’s pharmacy distribution sector is experiencing significant momentum. This growth springs from several powerful drivers: the nation’s escalating healthcare requirements, a population increasingly knowledgeable about health options, and a remarkably varied product spectrum. This spectrum spans critical long-term medications for chronic conditions right through to readily accessible over-the-counter wellness solutions. However, navigating this promising landscape carries inherent risks. Operating without robust regulatory adherence exposes even the most ambitious distributor pharmacy venture to severe consequences. Potential outcomes include substantial financial penalties, disruptive and costly product recalls, or, in the most drastic scenarios, a complete operational shutdown. The stakes are undeniably high.
For every player in this field – whether an independent pharmacy distributor, a larger-scale pharmacy wholesale distributor, or any entity functioning as a pharmacy distributor Malaysia relies on – aligning their operations with core compliance regulations isn’t merely a box-ticking exercise. It’s fundamentally vital for survival and sustainable growth. Importantly, this article delves into the complete regulatory structure governing pharmacy distribution service in Malaysia. We’ll provide straightforward insights into critical areas: securing the necessary licenses, obtaining essential NPRA (National Pharmaceutical Regulatory Agency) product approvals, implementing rigorous Good Distribution Practice (GDP) standards, and meeting the operational mandates governing every step, from initial importation right through to the final retail point. Crucially, understanding this framework is the first step towards resilience.

Ever wonder who keeps Malaysia’s medicine supply safe and reliable? It’s not just one group, but a coordinated framework ensuring every pill and potion meets strict standards before reaching patients. Understanding this oversight is crucial for anyone involved in healthcare.
The primary regulator is the National Pharmaceutical Regulatory Agency (NPRA), operating directly under the Ministry of Health (MOH). Working hand-in-hand with them is the Pharmacy Board Malaysia. Together, these bodies hold responsibility for the entire lifecycle of pharmaceuticals. Their critical tasks include vetting and approving new products (registration), issuing operating licenses, continuously monitoring drug safety, and rigorously enforcing Good Distribution Practice (GDP) standards nationwide. This comprehensive oversight is the bedrock of a trustworthy system.
This regulatory environment is defined by key pieces of legislation:
The Poisons Act 1952: This foundational law specifically governs the importation, sale, and overall distribution of substances classified as scheduled poisons. It provides the essential controls for higher-risk medications.
Control of Drugs and Cosmetics Regulations (CDCR) 1984: A much broader set of rules covering mandatory product registration, the licensing requirements for various operators, and strict guidelines for product labeling. This applies equally to both pharmaceutical items and cosmetics sold within Malaysia.
These regulations profoundly impact every single step within the pharmacy distribution chain operating here. Let’s look at concrete examples:
Mandatory Registration: Distributors are legally permitted only to handle pharmaceutical products that possess a valid MAL registration number, proving NPRA approval.
Strict Storage & Transport: Maintaining product integrity is non-negotiable. Medicines absolutely must be stored and shipped under meticulously controlled environmental conditions (like specific temperature and humidity ranges) throughout their entire journey.
Licensing & GDP Compliance: Every single business operating as a pharmacy wholesale distributor requires an official license. Beyond that, they must demonstrably adhere to comprehensive Good Distribution Practice (GDP) protocols covering everything from facility standards to staff training and documentation.
Enforcement isn’t just theoretical. The NPRA actively ensures compliance through a combination of regular audits of facilities and records, targeted enforcement raids where violations are suspected, and tight controls on application processes. This multi-pronged approach exists for one fundamental reason: safeguarding public health by guaranteeing safety and fostering unwavering trust across the entire pharmaceutical supply chain. Ultimately, knowing who sets the rules and how they work explains why Malaysia’s medicine distribution system functions with such essential reliability.

Before any company can legally distribute medicines or related products within Malaysia, securing specific licenses from the Ministry of Health’s Pharmaceutical Services Division (PSD) is absolutely non-negotiable. Understanding which permits apply to your operation is crucial for compliance and avoiding costly disruptions.
Malaysia’s regulatory framework primarily distinguishes between two critical license categories:
Type A License: This authorization is essential if your business activities involve the sale or supply of any substance classified as a poison or a controlled drug. It specifically governs these higher-risk categories.
Type B License: Required for businesses engaged in the manufacturing or wholesale distribution of pharmaceutical products that are already registered with the Malaysian authorities. This covers the bulk of standard medicine distribution channels.
Obtaining these licenses involves a structured process. Businesses must first formally register their entity with the Companies Commission of Malaysia (SSM). A foundational requirement is appointing a qualified, registered pharmacist who will take professional responsibility for overseeing pharmaceutical activities and ensuring regulatory adherence.
The application itself must be submitted electronically through the official Quest3+ NPRA online portal managed by the National Pharmaceutical Regulatory Agency. Beyond paperwork, your physical operations face significant scrutiny. Warehouse facilities and all storage areas must demonstrably comply with Good Distribution Practice (GDP) guidelines, focusing on product integrity, and meet stringent security protocols to prevent theft or diversion.
It’s vital to note that regulatory obligations apply equally across the board. Independent distributors, often managing smaller product volumes, operate under the exact same licensing rules and quality expectations as large chain-based distributor pharmacies. There is no relaxation of standards based on company size.
During mandatory licensing inspections, officers meticulously evaluate several key areas. They review comprehensive product documentation for accuracy and traceability. Storage conditions, inventory management systems, and record-keeping practices are thoroughly assessed. Furthermore, staff competency regarding relevant Standard Operating Procedures (SOPs) is verified. Falling short in any of these inspection criteria can actually result in significant application delays or outright denial of the license, potentially derailing your entire distribution schedule and market entry plans.

Think of Good Distribution Practice (GDP) as the essential shield protecting pharmaceutical products as they journey through the complex distribution maze. It’s not merely a formality; it’s the rigorous system ensuring medicines maintain their critical quality, safety, and integrity every step of the way – from the manufacturer’s door right down to the patient. In Malaysia, achieving GDP compliance isn’t optional; it’s a fundamental requirement. The National Pharmaceutical Regulatory Agency (NPRA) strictly enforces these standards, which carefully take cues from the broader ASEAN GDP Guidelines to ensure regional harmony.
So, what are the absolute pillars of GDP that distributors must nail? It starts with rock-solid environmental controls. This means meticulous temperature management throughout storage and transit, with robust cold chain monitoring systems mandatory for sensitive products like vaccines or biologics. Next up is impeccable physical handling – products need secure, appropriate packaging and careful movement procedures to prevent any damage or contamination. Crucially, comprehensive documentation forms the backbone of traceability: detailed records covering shipping invoices, precise batch numbers, and full shipment tracking history are non-negotiable. Finally, it’s about continuous improvement through regular internal audits and swift implementation of corrective actions whenever gaps are spotted.
For pharmacy wholesale distributors operating in Malaysia, proving ongoing GDP compliance isn’t a one-time thing. Concrete evidence must be submitted alongside both initial license applications and critical renewal submissions. Recognizing the stakes, leading Malaysian distributors proactively invest in key technologies: deploying digital data loggers for real-time temperature tracking, implementing sophisticated barcode or RFID systems enabling seamless stock traceability from warehouse to customer, and committing to thorough, ongoing staff training programs to drastically minimize risks associated with manual handling errors.
Falling short on GDP isn’t just a minor slip-up; it carries serious weight. Consequences can escalate quickly from costly product recalls that erode trust, to significant reputational damage that’s hard to recover from, and even potentially crippling restrictions on vital import or export activities. Getting GDP right is genuinely foundational for safe, reliable medicine supply. It’s definitely more than just paperwork.
Learn More : WHO: Guidelines – Distribution of health products

So your pharmaceutical shipment has successfully docked at a Malaysian port. That’s a significant milestone, but it’s far from the finish line. A complex web of regulatory and logistical steps unfolds next, crucial for ensuring product safety, efficacy, and legal compliance before those medicines reach pharmacies or hospitals. Understanding this post-arrival phase is vital for any importer.
First and foremost, all pharmaceutical products entering Malaysia require valid import permits. This isn’t optional. The foundational requirement, however, happens long before the ship sails: every single product must be officially registered with the National Pharmaceutical Regulatory Agency (NPRA). Successful registration earns the product its unique identifier, the essential MAL number. Without this number prominently displayed, clearance is impossible.
Importers bear the responsibility for navigating this process. Applications are managed electronically through the government’s designated Quest3+ system. Accuracy is paramount here. Submitted documentation must go beyond basic customs forms; it needs to include precise batch numbers and verifiable certification regarding the product’s origin. Missing or incorrect details can cause significant delays at this critical juncture.
Once cleared through initial customs, products move to storage facilities. This is where strict adherence to labeled storage conditions becomes non-negotiable. Warehouse temperatures must be meticulously maintained exactly as specified on each product’s packaging. Deviations risk compromising the medicine’s integrity.
Furthermore, robust inventory management is essential. Facilities must have clear procedures to physically segregate any stock that is expired, damaged, or subject to a recall. This prevents accidental distribution of compromised goods. For sensitive items like vaccines or biological therapies, the standards are even higher. These require dedicated refrigeration systems equipped with continuous temperature monitoring and audible alarms to alert staff instantly to any fluctuations. The stakes are simply too high for passive monitoring.
Malaysia’s tropical climate presents a persistent challenge. Intense heat and humidity aren’t just uncomfortable; they’re a major threat to pharmaceutical stability. For biologics, certain supplements, and many standard medicines sensitive to heat, even a brief failure in warehouse cooling systems can lead to catastrophic losses, potentially ruining an entire shipment. Investing in reliable, validated cold chain infrastructure isn’t just best practice; it’s a fundamental business necessity in this environment.
Given these complexities, navigating the journey from port arrival to the final pharmacy shelf demands expertise. This is why successful pharmaceutical distributors frequently collaborate with specialized logistics providers who hold Good Distribution Practice (GDP) certification. These partners understand the stringent requirements for handling pharmaceuticals throughout the supply chain. They provide the assurance of end-to-end compliance, managing temperature-controlled transportation, secure warehousing, and meticulous documentation – ensuring the product’s safety and efficacy is preserved right up to the point it reaches the patient. Getting to port is step one; navigating what comes after determines real success in the Malaysian market. It’s a process where cutting corners simply isn’t an option.
Learn More : The Role of Third‑Party Logistics (3PL) in Malaysia’s Pharmaceutical Supply Chain | Good Distribution Practice | European Medicines Agency

Ever wonder how consumers can genuinely trust a product’s journey, confident it’s authentic from factory floor to retail shelf? In Malaysia, the cornerstone of combating counterfeits hinges on robust product traceability systems. Current regulations meticulously outline this framework, demanding transparency at every step to safeguard public health and market integrity.
At the heart of the strategy lies mandatory identification. Every single registered product circulating within Malaysia must visibly display its unique MAL number, a crucial identifier issued by the National Pharmaceutical Regulatory Agency (NPRA). Alongside this, clear manufacturer details are obligatory – no anonymity allowed. Furthermore, for items deemed high-risk, such as certain pharmaceuticals or critical goods, serialization is becoming the expected standard. This means assigning unique, scannable codes to individual units or batches, creating an unforgeable digital fingerprint. Complementing these identifiers is the mandate for comprehensive record retention. Businesses must securely maintain detailed transaction and handling logs for a minimum of five years, ensuring a verifiable audit trail is always accessible if questions arise.
Distributors aren’t just middlemen; they shoulder immense responsibility in upholding this traceability shield. Their pivotal duties include meticulously documenting batch numbers upon both receiving goods from manufacturers and dispatching them further down the line. This accurate record-keeping is fundamental for pinpointing specific products should issues emerge. Speed is also paramount: distributors must be primed to execute recalls within hours, not days, minimizing potential public risk. Vigilance against parallel import errors is equally vital; unintentionally handling non-compliant imports can trigger significant enforcement actions and undermine the entire system’s credibility.
Recognizing the complexity of modern supply chains, many forward-thinking distributor pharmacies are adopting sophisticated inventory tracking software. These powerful platforms often integrate directly with NPRA databases, creating a seamless flow of verification data. This tech integration enables near-instantaneous validation of product legitimacy and provides real-time traceability. A quick scan can confirm a product’s registration status, batch history, and movement, offering unprecedented peace of mind to both businesses and, ultimately, the end consumer.
Ultimately, Malaysia’s approach combines clear regulatory mandates with critical distributor accountability, increasingly supercharged by digital solutions. This multi-layered system – anchored by the MAL number, enforced record-keeping, serialization, and tech-enabled checks – builds a formidable defense against fakes, proving a product’s legitimacy every step of the way. It’s about creating an environment where trust is verifiable, not just assumed.
Learn More : The Role of Pharmacy Distribution Services in Malaysia’s Healthcare System
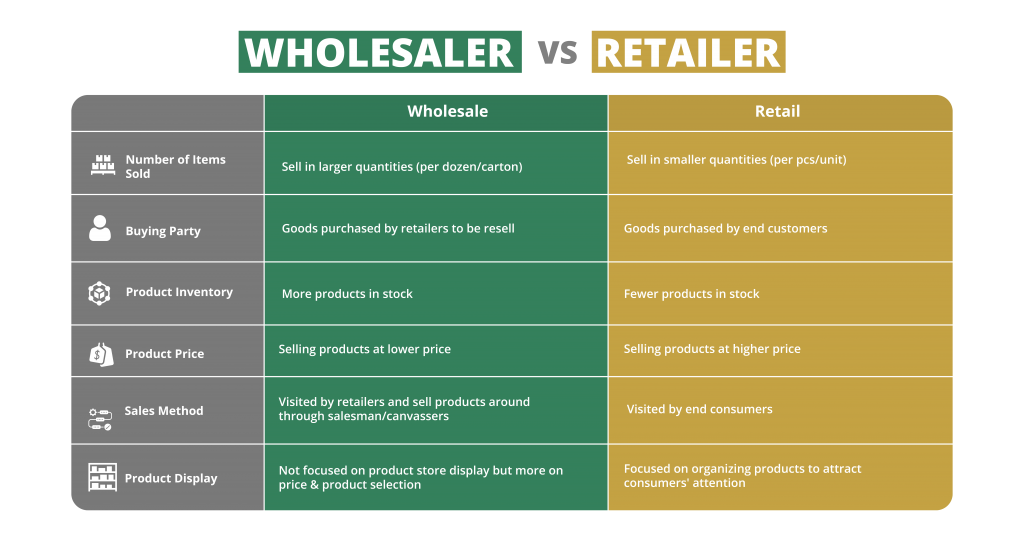
Here’s the reality: compliance obligations aren’t one-size-fits-all in the pharmacy world. They fundamentally shift based on whether you’re serving patients directly or supplying other businesses. Understanding this distinction is crucial for navigating the regulatory landscape effectively.
If you operate a retail pharmacy, your primary interaction is with consumers. This model brings specific, patient-centric burdens. You absolutely must employ state-licensed pharmacists to oversee operations and ensure patient safety. Adhering meticulously to labeling standards – clear dosage instructions, warnings, expiration dates – is non-negotiable. Providing appropriate patient counseling for prescriptions, especially new medications or complex regimens, is another core requirement. Furthermore, robust systems for tracking every single prescription, with heightened vigilance for controlled substances and other scheduled drugs, form the backbone of your compliance efforts. Missing any of these can land you in serious trouble.
Operating as a wholesale pharmacy distributor means your customers are other licensed entities, not the public. Your compliance focus pivots sharply towards supply chain integrity and product handling. Strict adherence to Good Distribution Practices (GDP) for all pharmaceutical stock is paramount – this governs storage conditions, transportation security, and overall product handling to prevent contamination or diversion. Maintaining comprehensive transaction records is essential for complete traceability, allowing regulators to track a product’s journey from manufacturer to endpoint. Expect regular, often unannounced, inspections of your warehouse facilities and logistics processes by authorities. These are pretty strict.
Chain retailers operating their own distribution centers face a double layer of complexity. They must diligently manage both the retail compliance requirements and the wholesale distributor standards internally. Conversely, independent pharmacy distributors might choose to outsource certain logistics functions like warehousing or transportation to third-party logistics providers (3PLs). However, crucially, the ultimate accountability for maintaining accurate documentation and ensuring overall regulatory compliance always rests squarely with the distributor licensee. You can’t outsource responsibility.
Regardless of your business model, being perpetually prepared for audits by the Ministry of Health (MOH) or other regulators is non-negotiable. This readiness hinges on several key elements: having meticulously detailed Standard Operating Procedures (SOPs) readily accessible, maintaining up-to-date logs proving equipment calibration (think refrigerators, scales), and keeping thorough records demonstrating how product quality complaints were investigated and resolved. Investing time and resources into robust audit preparation isn’t just about checking boxes; it directly translates to significant savings in both time and money when inspectors inevitably arrive. Being unprepared is simply too costly.

Operating within Malaysia’s tightly regulated pharmaceutical sector demands strict adherence to all protocols. Deviating from the required steps isn’t just risky; it invites serious, costly repercussions from the National Pharmaceutical Regulatory Agency (NPRA). Understanding these consequences is vital for any distributor or manufacturer aiming for sustainable operations.
The NPRA wields significant authority to penalize non-compliance, and the financial and operational hits can be severe. Companies caught distributing products without proper authorization face substantial monetary fines – these can soar as high as RM50,000 per violation. But the pain doesn’t stop there. Regulatory bodies possess the power to suspend, or even permanently revoke, a company’s crucial operating licenses, effectively halting business. Further enforcement actions include the outright seizure of non-compliant goods and the damaging prospect of being blacklisted within the industry supply chain. These aren’t just hypothetical risks; they’re enforceable realities.
Real-world examples underscore the NPRA’s active vigilance. Just recently, a distributor based in Johor faced significant fines after being caught selling dietary supplements that lacked mandatory registration. In another notable operation, NPRA officials conducted a targeted raid on a storage facility in Klang. This warehouse was found holding expired medicinal products under conditions that dangerously neglected essential cold chain temperature controls – a critical violation for product safety.
The NPRA isn’t relying solely on physical inspections. Their adoption of a sophisticated digital reporting platform has significantly boosted enforcement capabilities. This system allows for dramatically faster initiation of investigations and crucially facilitates anonymous tip-offs from whistleblowers and industry insiders. The result? A measurable increase in detection rates and swifter action against violators.
What typically draws the NPRA’s scrutiny? Audits are frequently prompted by several recurring issues. Incomplete or missing batch manufacturing records are major red flags, as they hinder product traceability. Failures in maintaining the unbroken cold chain – essential for temperature-sensitive medicines – consistently attract regulatory attention. Furthermore, operating storage facilities without the necessary NPRA licenses remains a primary trigger for enforcement action. Staying compliant means proactively addressing these critical areas.
Learn More : An Early‑Warning System Will Make Your Supply Chain More Resilient
For Malaysian pharmacy distributors, navigating the tightrope between rapid growth and strict regulatory compliance can feel impossible. Slowing down isn’t an option, but neither is cutting corners with bodies like the NPRA watching closely. So, how do you scale effectively without tripping over compliance hurdles? The answer lies in weaving regulatory adherence directly into your operational DNA, not treating it as a last-minute box to tick.
Simply reacting to rules won’t cut it. Sustainable growth demands proactive strategies:
Crystal-Clear Procedures: Don’t leave things to chance. Develop detailed, unambiguous Standard Operating Procedures (SOPs) covering every critical touchpoint – storage conditions, delivery protocols, and especially recall processes. Everyone needs to know the exact steps.
Empower Your People: Your logistics and admin teams are the front line. Invest seriously in ongoing, targeted compliance training. Make sure they truly understand why procedures exist and the consequences of lapses; knowledge is their best tool.
Embrace Digital Oversight: Ditch manual logs prone to error. Implement robust digital inventory management systems featuring comprehensive batch tracking. This isn’t just efficient; it provides an undeniable audit trail for regulators.
Choose Partners Wisely: Your logistics provider’s standards reflect on you. Insist on partnering only with GDP-certified (Good Distribution Practice) specialists. Their commitment to compliance directly safeguards your products’ integrity during transit.
Technology is a powerful ally in balancing speed and safety:
QR Code Tracking: Implement QR-based systems for instant product traceability. Scanning a code reveals a product’s entire journey, massively speeding up recalls or verification checks.
Real-Time Environmental Monitoring: Warehouse temperature and humidity are non-negotiable. Use connected dashboards providing live monitoring and alerts, ensuring your storage environment always meets requirements without constant manual checks.
Streamlined Internal Audits: Don’t reinvent the wheel for audits. Utilize internal checklists meticulously aligned with the latest NPRA standards. This ensures consistent self-assessment and identifies potential gaps before they become external findings.
The most successful distributors here in Malaysia understand this deeply: compliance isn’t a separate task or a growth barrier. It’s an essential, integrated system within the business itself. They build it into their processes from day one. In fact, leading players often appoint dedicated compliance officers. These professionals don’t just react; they proactively oversee audits, prepare meticulous reports, and constantly ensure operations align seamlessly with regulations. This proactive stance transforms compliance from a cost center into a genuine competitive advantage, enabling smoother, faster, and ultimately sustainable growth.
Learn More : Why Efficient Pharmacy Distribution is Crucial for Malaysia’s Rural Healthcare Development | A Guide to Good Distribution Practice (GDP)
Let’s be honest: in the fiercely competitive world of pharmaceutical distribution, merely ticking regulatory boxes isn’t enough anymore. True compliance? It’s become the undeniable mark of a distributor that’s credible, genuinely capable, and deeply cares about patient safety. This is especially critical navigating Malaysia’s intricate pharmaceutical ecosystem, where regulations are constantly evolving. Distributors who truly get it – who master NPRA approval workflows, operate with unwavering GDP certification, and proactively invest in advanced traceability tech – aren’t just surviving; they’re building resilient, trusted businesses positioned for real growth.
So, what does building that unshakeable compliance foundation actually involve? It’s a continuous commitment woven into every operational thread:
Licensing is Non-Negotiable: Securing the correct NPRA licenses – whether it’s the crucial Type A or Type B – forms the absolute bedrock of your legal operation. Don’t guess on this one.
Guidelines as Gospel: Following NPRA regulations and Good Distribution Practice (GDP) standards meticulously isn’t optional paperwork; it’s the operational blueprint for safety and quality every single day.
Integrity in Storage & Transport: Maintaining rigorously controlled, temperature-assured environments throughout the entire supply chain journey – from warehouse shelf to pharmacy door – is paramount for product efficacy.
Traceability You Can Trust: Implementing robust, end-to-end traceability systems ensures you can track any product’s journey instantly. This isn’t just for audits; it’s vital for swift recalls and patient protection.
Audit Readiness, Always: Treating compliance as an everyday practice, not a last-minute scramble, means you’re confidently prepared for any NPRA audit, anytime they walk through the door.
Who Needs This Edge? Frankly, Everyone in the Chain:
Are you an independent pharmacy distributor aiming to scale securely?
Is your retail pharmacy brand strategically expanding into wholesale distribution channels?
Maybe you’re a logistics partner identifying where compliance gaps might risk client relationships?
Forget seeing regulations as a burden. Staying demonstrably ahead of NPRA requirements is no longer optional overhead; it’s your most powerful competitive differentiator in the Malaysian market. It builds trust with regulators, partners, and ultimately, the patients relying on those vital medicines.
Whether you’re meticulously planning the launch of a new pharmacy distribution venture in Malaysia or conducting a thorough review of your existing logistics and compliance practices, PriooCare offers more than just advice. We provide strategic partnership. Our tailored support covers the full spectrum: navigating complex licensing (Type A/B), implementing watertight GDP protocols, ensuring seamless regulatory readiness, and even optimizing merchandising solutions designed to accelerate your brand’s specific growth trajectory. Don’t just react to regulations – lead because of them.