
September 11, 2025
For pharmacy distributors in Malaysia, the NPRA registration fees aren’t just another line item in the budget—they’re a strategic factor that shapes market entry, cash flow, and long-term competitiveness. The National Pharmaceutical Regulatory Agency (NPRA) serves as the gatekeeper for product safety, ensuring only compliant pharmaceuticals, supplements, and cosmetics reach consumers. Underestimating these costs can lead to delayed launches, regulatory penalties, or even failed product distribution—mistakes that erode trust and profitability.
Consider this real-world scenario: A Kuala Lumpur-based distributor rushed a new immune-boosting supplement to market without accounting for the RM3,000–RM5,000 evaluation fee. Halfway through the process, cash flow gaps forced them to pause, missing the seasonal demand window. Competitors with reliable fee planning captured the market instead.
Data reinforces the stakes:
72% of regulatory delays trace back to fee-related miscalculations (Malaysian Pharmaceutical Society, 2023).
Products with accurate NPRA cost projections see 40% faster time-to-market versus industry averages.
The practical implication? Treating NPRA fees as an afterthought risks inventory bottlenecks, inflated retail pricing, and eroded margins—especially for high-volume categories like health supplements or cosmetics. For licensed pharmacy wholesale distributors, mastering this fee structure isn’t optional; it’s the proven backbone of efficient operations.

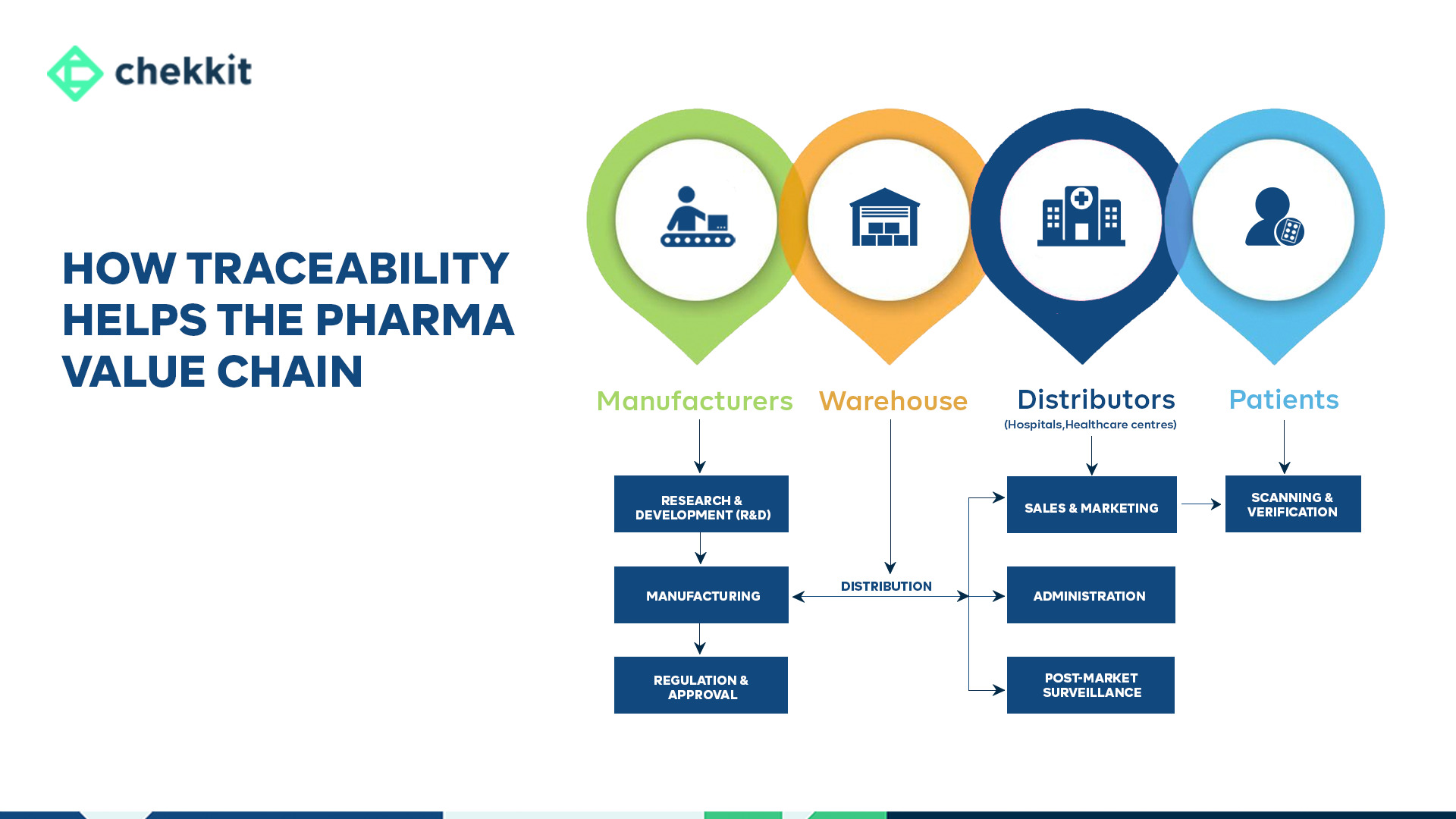
What makes the NPRA such a pivotal player in Malaysia’s healthcare logistics? Operating under the Ministry of Health (MOH), this agency oversees everything from clinical evaluations to post-market surveillance—ensuring every therapeutic product, from prescription drugs to herbal tonics, meets stringent safety benchmarks.
Here’s why pharmacy stakeholders can’t afford to overlook its influence:
Market Access Control: Without NPRA approval, even the most effective products sit in warehouses. A Johor Bahru distributor learned this the hard way when a batch of OTC pain relievers was rejected over incomplete stability data—a 6-month setback.
ASEAN Alignment: NPRA’s digital transformation (e.g., Quest 3+ online submissions) syncs with regional standards, cutting approval times by 30% for compliant dossiers.
Anti-Counterfeit Shield: The agency’s track-and-trace protocols protect distributor pharmacies from sourcing fraudulent inventory—a growing issue in Penang and Selangor, where counterfeit vitamin supplements surged 22% in 2022.
For pharma distributors, NPRA compliance isn’t just red tape; it’s a trusted badge of credibility. Brands partnering with expert distributors leverage this for strategic advantages—faster shelf placement, stronger retailer negotiations, and repeat orders.
Learn More : KKM Approval Malaysia: Legal Requirements for Sellers
Assuming NPRA fees are uniform across product categories is a costly misstep. The agency tailors costs based on risk, formulation complexity, and regulatory scrutiny—a nuance that separates prepared distributors from those bleeding profits.
Breakdown by product type:
Prescription (Scheduled Poisons):
Screening: RM1,000
Evaluation: RM6,000–RM10,000 (varies by molecule novelty)
Non-prescription (General Sales):
Screening: RM800
Evaluation: RM4,000–RM6,000
Example: A Kuching-based distributor importing diabetes meds budgeted RM8,000 for evaluation but missed the RM1,200 stability study add-on—a 14% cost overrun.
Screening: RM600–RM1,000
Evaluation: RM3,000–RM5,000 (higher for multi-ingredient blends)
Malaysian trend alert: Collagen peptides and black seed oil capsules dominate registrations, with 1,200+ applications in 2023 alone.
Notification fee: RM200–RM500 (lower scrutiny, but post-market audits apply)
Operational tip: A Selangor skincare distributor slashed costs by 15% grouping similar products (e.g., serums + moisturizers) under one dossier.
Comparative insight:
| Category | Screening Fee | Evaluation Fee |
|---|---|---|
| Prescription Drugs | RM1,000 | RM6,000–RM10,000 |
| Health Supplements | RM600–RM1,000 | RM3,000–RM5,000 |
| Cosmetics | RM200–RM500 | Not applicable |
Learn More : WHO Guidelines on Registration of Medicines
-1.png?width=1584&height=960&name=Axify%20blogue%20header%20(6)-1.png)
The NPRA registration process unfolds in phases—each with its own cost and compliance demands. Missteps at any stage trigger expensive rework.
Administrative Screening (RM500–RM1,000)
Purpose: Verify document completeness (e.g., GMP certificates, labeling drafts).
Pitfall: A Kuala Lumpur importer misclassified a probiotic supplement as a “traditional medicine,” incurring RM2,000 in re-submission fees.
Dossier Evaluation (RM3,000–RM10,000)
Purpose: Scientific review of efficacy, safety, and manufacturing controls.
Pro tip: 80% of rejections link to incomplete stability data—budget RM3,500–RM5,000 for third-party testing.
Renewal & Variations (60–70% of initial cost)
Renewals every 5 years are non-negotiable.
Variations (e.g., new flavor, dosage form) cost RM500–RM2,000.
Checklist for smooth processing:
✅ Pre-submit a document gap analysis
✅ Allocate 10–15% buffer for unexpected charges
✅ Track NPRA’s quarterly fee updates (e.g., 2024’s 4% hike for biologics)
Learn More : ASEAN Harmonization of Pharmaceutical Regulations

Imported products face higher NPRA costs—a strategic consideration for pharmacy wholesale distributors sourcing overseas.
Key differences:
Local products: Lower fees due to pre-vetted GMP compliance.
Imported goods: Added costs for:
Document translation (RM200–RM500 per dossier)
GMP equivalency checks (RM300–RM700)
Climate stability data (RM1,500+ for tropical countries)
Malaysian case study: A Penang distributor importing German wound care gels paid RM1,200 extra for EU-to-Malaysia storage condition validations—a cost passed to hospitals via 8% higher unit pricing.
Framework for cost mitigation:
Negotiate with suppliers to share dossier preparation costs.
Batch submissions for similar products (e.g., all vitamin D variants).
Leverage ASEAN harmonization for brands with existing Thai or Singapore approvals.

The NPRA registration fees are just the tip of the iceberg for pharmacy distributors—what follows are ripple effects that reshape supply chain dynamics, cash flow, and market entry strategies. A Klang Valley-based distributor learned this the hard way when a delayed approval for a popular probiotic brand forced them to hold RM500,000 worth of stock for 4 extra months, incurring RM35,000 in unplanned storage fees.
Operational impacts breakdown:
Stock Planning : Products stuck in regulatory limbo can’t generate revenue. Distributors juggling 50+ SKUs often face 3–6 month delays, derailing promotions tied to festive seasons (e.g., Raya health supplement campaigns).
Cash Flow Strain : Upfront fees (e.g., RM10,000 for prescription drugs) + holding costs squeeze margins. A Penang distributor reported 12% lower profitability on imported skincare due to unbudgeted dossier amendments.
Market Timing Risks : Launching a diabetic care product post-approval but after a competitor’s campaign can slash 30–40% of projected sales.
Proven workaround :
Integrate NPRA timelines into ERP systems to auto-adjust purchase orders and marketing spends. For health supplements, buffer 6–8 months lead time—twice the duration for cosmetics.
Learn More : Pharmaceutical Retail Merchandising
![]()
Top-tier pharma distributors treat NPRA fees like chess moves—anticipating costs 3 steps ahead. Here’s how trusted players like Caring Pharmacy’s suppliers optimize planning:
Strategic Framework :
Fee Matrix Mapping
Maintain a live database of NPRA fees (e.g., 2024’s 10% hike for biologics).
Example: A Johor Bahru importer saved RM28,000/year by switching from single-dossier to bulk submissions for similar supplements.
SKU Forecasting
Embed NPRA costs into per-unit economics. For RM5,000 evaluation fees on a RM15/unit supplement, sell minimum 1,000 units to break even.
Co-Sharing Models
Negotiate 60/40 splits with brands for exclusive distribution rights. A Kuching distributor cut 32% of registration costs this way for Australian vitamins.
Checklist for Agile Budgeting :
✅ Assign a regulatory cost code in accounting software
✅ Quarterly reviews of NPRA circulars for fee updates
✅ Build 5%–8% contingency for hidden costs (e.g., GMP audit surcharges)
Learn More : How Regulatory Compliance Affects Pharmaceutical Supply Chains
Malaysia’s regulatory landscape is shifting—digitally and financially. Ignoring these changes risks turning efficient distributors into reactive ones.
2024’s Critical Updates :
Digital-Only Submissions : The Quest3+ portal slashes processing time by 25% but requires PDF/XLS templates—a hurdle for older distributors in East Malaysia.
Fee Increases : NPRA’s 10% across-the-board hike (2023) impacts high-volume categories like OTC medicines (now RM4,400–RM6,600 per evaluation).
Fast-Track Pathways : Critical care drugs (e.g., oncology meds) now get 30-day approvals if aligned with MOH’s National Formulary.
Local Adaptation :
A Kuala Lumpur startup importing Korean cosmetics leveraged digital submission to cut registration time from 90 to 47 days—outpacing competitors still using paper dossiers.
Monitoring Tools :
| Resource | Frequency | Key Use |
|---|---|---|
| NPRA Website | Weekly | Fee revisions, new guidelines |
| MOH Quarterly Bulletins | Monthly | Policy shifts (e.g., ASEAN harmonization) |
| MPC Webinars | Bi-annually | Best practices for SMEs |
Learn more : Trends in Health Product Regulation in Asia

Even expert distributors stumble on NPRA fee traps. A Malacca-based company lost RM120,000 resubmitting a traditional medicine line misclassified as health supplements—a 100% avoidable error.
Top 3 Pitfalls & Fixes :
Hidden Costs Blindspots
Issue: Forgetting RM200–RM500/document translation or RM1,500/stability studies.
Fix: Use a pre-submission calculator (e.g., NPRA’s Fee Estimator Tool).
Misclassification Mishaps
Issue: Registering CBD oils as health supplements (banned) vs. controlled drugs.
Fix: Request NPRA’s pre-classification guidance (takes 5 business days).
Renewal Amnesia
Issue: Letting a top-selling vitamin’s registration lapse, triggering delisting.
Fix: Set Google Calendar alerts for 4.5-year marks (6-month renewal buffer).
Learn More : Top Mistakes When Applying for KKM Approval
Mastering NPRA fee structures isn’t about compliance alone—it’s about transforming regulatory hurdles into competitive leverage. Whether you’re a nicure supplement importer in Ipoh or a multinational pharma distributor in Cyberjaya, the rules are the same:
Strategic forecasting beats reactive scrambling.
Digital adoption (e.g., Quest3+) is now non-negotiable.
Local partnerships (e.g., regulatory consultants) unlock faster approvals.
The proven distributors thrive by treating NPRA fees as investments, not expenses—tailoring each submission to Malaysia’s evolving healthcare logistics.
Q1: How to check NPRA product search?
Answer:
You can use the NPRA Product Search via the NPRA official website. Go to the “Product Registration” section, enter the product name or registration number (MAL code), and review the official status listed in the database.
Q2: How can I verify a product’s NPRA KKM registration?
Answer:
Check the product’s MAL number (e.g., MAL12345678T) and search it on the NPRA database. A valid product will show the registration holder, manufacturer, classification, and approval status.
Q3: What is NPRA?
Answer:
NPRA stands for the National Pharmaceutical Regulatory Agency, the Malaysian authority responsible for regulating the quality, safety, and efficacy of medicines, cosmetics, supplements, and health products.
Q4: What products need a KKM check?
Answer:
Any product claiming therapeutic effects—such as medicines, supplements, traditional products, medical devices, and cosmetics—must undergo a KKM check to ensure they are registered and approved by NPRA.
Q5: What is NPRA KKM?
Answer:
NPRA KKM refers to the National Pharmaceutical Regulatory Agency under the Ministry of Health (KKM). It manages product registration, safety monitoring, and regulatory compliance for health-related products in Malaysia.
Q6: How to get KKM approval for a product?
Answer:
You must apply through the NPRA QUEST system, submit required documentation, provide product safety and quality data, pay the necessary fees, and wait for NPRA evaluation before approval is granted.
Q7: What is a KKM-approved product?
Answer:
A KKM-approved product is one that has passed NPRA’s safety, quality, and efficacy assessments and has been issued a valid MAL registration number for sale in Malaysia.
Q8: What is NPRA for?
Answer:
NPRA oversees the regulation of pharmaceuticals, cosmetics, traditional medicines, and health products to ensure public safety. It evaluates product applications, monitors adverse events, and enforces compliance standards.
Q9: How to register NPRA in Malaysia?
Answer:
Companies must create a QUEST account, prepare technical documents (COS, GMP, test reports), submit an application for product registration, and follow NPRA’s evaluation process until approval is granted.
Q10: How do I check the status of my NPRA product?
Answer:
Log in to your NPRA QUEST account and view your submission dashboard. You can check whether your product is under evaluation, queried, approved, or rejected. The public can also check via the NPRA Product Search database.
📞 Let’s Talk: Need a Regulatory or Distribution Partner?
Navigating Malaysia’s pharmacy distribution landscape requires trusted expertise. At PriooCare, we specialize in:
End-to-end NPRA registration support
Cost-optimized launch planning
In-pharmacy merchandising strategies
Reach out today to turn regulatory challenges into scalable growth.
Our marketing and sales teams use their strong relationships with the channel to create demand for your product at every stage of its lifecycle.
Demand creation services we offer: