
September 10, 2025
In an era where counterfeit health products and unregistered supplements flood the market, Malaysian pharmacies face mounting pressure to ensure every item on their shelves meets strict regulatory standards. The consequences of failing to verify product legitimacy? Severe fines, reputational damage, and even license revocation—risks no pharmacy distributor Malaysia-based business can afford.
Consider this real-world scenario: A well-known independent pharmacy distributor in Kuala Lumpur faced a public backlash after a customer reported adverse effects from an unregistered skin-lightening cream. The product, sourced from an unauthorized supplier, lacked NPRA approval, leading to a forced recall, regulatory penalties, and a 30% drop in customer trust within weeks.
72% of Malaysian consumers actively check for NPRA registration before purchasing health supplements (MOH Survey, 2023).
Over 150 product seizures occurred in 2022 alone due to non-compliance (NPRA Enforcement Report).
For pharmacy wholesale distributors, this isn’t just about compliance—it’s about operational survival. Integrating NPRA product verification into procurement workflows is no longer optional; it’s a strategic necessity to mitigate risk and uphold brand integrity.

The National Pharmaceutical Regulatory Agency (NPRA) isn’t just another bureaucratic body—it’s the cornerstone of pharmaceutical safety in Malaysia. Operating under the Ministry of Health (MOH), the NPRA’s role is non-negotiable: ensuring every medicine, supplement, and cosmetic product meets rigorous safety benchmarks before reaching consumers.
But why does this matter for distributor pharmacy operations?
Legal Mandate: Under the Sale of Drugs Act 1952, distributing unregistered products is a punishable offense—fines can exceed RM 50,000 per violation.
Consumer Trust: Patients rely on pharmacies to provide expert-vetted products. A single lapse can erode decades of credibility.
Supply Chain Integrity: Manufacturers with NPRA-approved facilities follow Good Manufacturing Practices (GMP), reducing contamination risks.
Active Pharmaceutical Ingredients (APIs): Verified for safety and efficacy.
Labeling Compliance: Claims must align with NPRA-approved indications (e.g., a “cholesterol-lowering” supplement must have clinical backing).
GMP Audits: Manufacturing sites undergo annual inspections.
Local Example: In Penang, a pharmacy distribution service avoided a regulatory shutdown by cross-checking a new herbal supplement’s MAL number before stocking it—discovering it had been delisted six months prior due to undisclosed steroids.
For independent pharmacy distributors, leveraging the NPRA portal isn’t just about avoiding penalties—it’s about strategic sourcing and long-term viability.

Imagine this: Your pharmacy wholesale distribution business receives a lucrative offer for a “miracle” pain relief cream at half the market price. Tempting? Absolutely. Risky? Catastrophically so.
Distributing unregistered products in Malaysia carries three immediate threats:
✅ Regulatory Fines
NPRA enforcement teams conduct random raids on warehouses and pharmacies.
Penalties range from RM 25,000 (first offense) to license suspension (repeat violations).
✅ Product Seizures
In 2023, NPRA confiscated over 12,000 units of unregistered erectile dysfunction drugs in Johor Bahru alone.
Losses include inventory costs, logistical disruptions, and legal fees.
✅ Brand Damage
A single viral post about “fake medicines” can tank customer loyalty.
Example: A Klang Valley pharmacy chain lost 40% of its corporate clients after a viral TikTok exposed unregistered baby formula on its shelves.
Cross-reference product name and MAL number on NPRA’s portal.
Verify the holder’s license status (avoid “ghost” manufacturers).
Check for expired registrations (even previously approved products lapse).
For distributor pharmacy networks, these steps aren’t just best practices—they’re business-preserving protocols.
Learn More : Pharmacovigilance Guidance Top Mistakes When Applying for KKM Approval | KKM Approval Malaysia: Legal Requirements for Sellers | Drug Registration Guidance Document
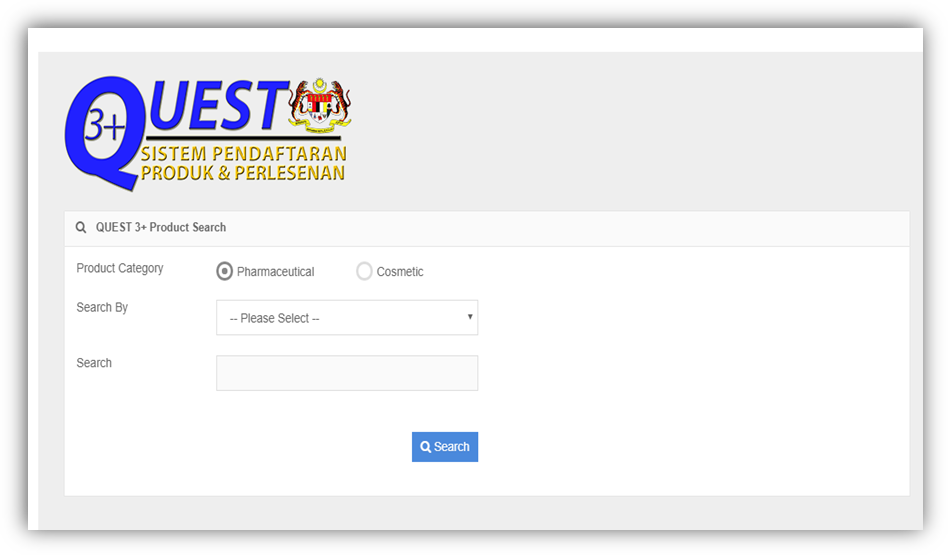
The NPRA product search tool is a game-changer for pharmacy distributors Malaysia-wide—but only if used correctly. Here’s a proven, efficient method to maximize its utility:
Visit npra.gov.my → Navigate to “QUEST3+ Registered Product Search”.
Product Name: Best for branded items (e.g., “Brand X Collagen”).
Registration Number (MAL): Instant validation (e.g., “MAL20034565X”).
Active Ingredient: Critical for generics (e.g., “paracetamol 500mg”).
Drug Class: “Scheduled Poison” vs. “Over-the-Counter (OTC)”.
Status: “Registered” vs. “Cancelled” (shockingly, 8% of listings expire annually).
Match the brand name, holder details, and expiry date.
Red Flag: Discrepancies in manufacturer address vs. supplier claim
Operational Scenario: A Kuching-based pharmacy distributor avoided a RM 100,000+ recall by noticing a skincare product’s “Cancelled” status—despite the supplier’s “valid until 2025” paperwork.
Why do NPRA search filters matter? Because generic searches waste time and increase oversight risks. Here’s how to use them strategically:
Cosmetics: Ensure labels don’t make unauthorized therapeutic claims (e.g., “treats eczema”).
Health Supplements: Verify vitamin dosages comply with NPRA’s daily limits.
Why It Matters: Some manufacturers operate multiple shell companies to bypass compliance.
Pro Tip: Cross-check the holder’s other registered products for consistency.
Use Case: A Selangor pharmacy identified a “new” flu medicine as a rebranded, deregistered product by searching its paracetamol + pseudoephedrine combo.
Comparison: NPRA vs. Supplier Documentation
| Criteria | NPRA Portal | Supplier Documents |
|---|---|---|
| Accuracy | Real-time, government-verified | Often outdated or forged |
| Liability | Official record | Self-reported (high risk) |
For independent pharmacy distributors, mastering these filters is essential to streamline audits and reduce procurement risks.

Running an NPRA product search is just the first step—deciphering the results is where the real compliance work begins. For pharmacy distributors Malaysia-wide, misreading even a single field can lead to costly mistakes, from stocking expired products to partnering with unverified suppliers.
Here’s what to scrutinize in every NPRA search result:
✅ MAL Number
Format: Must follow MAL[YEAR][XXXXX][Letter] (e.g., MAL202412345A).
Red Flag: Missing or malformed numbers (e.g., “MAL-24-1234”) often indicate fraudulent listings.
✅ Product Status
Only “Registered” status guarantees compliance.
Avoid: “Cancelled,” “Expired,” or “Pending” (even if the supplier insists it’s “under review”).
✅ Holder Name Alignment
Cross-check the license holder against your supplier’s documentation.
Local Example: A Johor Bahru independent pharmacy distributor discovered their “authorized” diabetes supplements were registered to a different company—a classic parallel import risk.
✅ Validity Period
NPRA registrations expire annually unless renewed.
Pro Tip: Set calendar reminders for 3 months before expiry to avoid last-minute delistings.
Why This Matters: In 2023, NPRA reported 23% of inspected pharmacies carried at least one product with lapsed registration—a preventable risk with diligent verification.

The NPRA portal isn’t just for emergencies—it’s a strategic asset for daily operations. Here’s how Malaysian pharma distributors leverage it:
Scenario: A Klang Valley distributor was offered a “FDA-approved” weight-loss tea at a 50% discount.
Action: NPRA search revealed the product’s MAL number was revoked due to undeclared sibutramine.
Outcome: Avoided a RM 200,000+ recall and potential MOH blacklisting.
Scenario: A Penang pharmacy chain used batch searches to audit 500+ SKUs.
Discovery: 7 cosmetic products showed “Cancelled” statuses despite being sold as “new arrivals.”
Solution: Immediate delisting + supplier contract termination.
Key Takeaway: Whether you’re a distributor pharmacy or a retail network, NPRA checks are your first line of defense against compliance breaches.

For pharmacy wholesale distributors, manual NPRA checks won’t scale. Here’s how top players bake compliance into their workflows:
Mandate MAL number submission for all new product pitches.
Require suppliers to provide NPRA registration certificates (not just invoices).
Assign a compliance officer to audit 10% of SKUs monthly.
ERP Integration: Sync NPRA’s database with procurement software to auto-flag unregistered products.
Local Success: A Sarawak-based distributor reduced compliance errors by 65% after embedding real-time MAL validation into their ordering system.
Conduct quarterly workshops on interpreting NPRA results.
Example: Teach teams to spot fake MAL numbers (e.g., duplicates, invalid prefixes).
Why It Works: Institutionalizing NPRA checks transforms compliance from a reactive chore into a proactive advantage.
Learn More : What is the Role of Merchandising? | Pharma Supply Chain Traceability

Beyond avoiding fines, NPRA product search fortifies the entire healthcare logistics ecosystem:
✅ Combats Counterfeits
Statistic: NPRA seizures of fake medicines rose by 18% in 2023—most from unauthorized online sellers.
✅ Streamlines B2B Trust
Suppliers with 100% NPRA-compliant catalogs gain preferred partner status with hospitals.
✅ Protects Patients
Real Impact: A Kuala Lumpur clinic avoided administering unregistered antibiotics by verifying every shipment.
Bigger Picture: Consistent NPRA use aligns with MOH’s 2025 agenda to eliminate substandard pharmaceuticals from Malaysia’s supply chain.
Learn More : Overview of ASEAN Regulatory Systems
For pharmacy distributors Malaysia relies on, NPRA verification isn’t optional—it’s the bedrock of sustainable operations.
Retailers using NPRA tools report fewer customer complaints and higher repeat purchases.
Wholesalers with automated checks gain faster MOH approvals for tender bids.
The Bottom Line: In an industry where trust is currency, NPRA compliance isn’t just about rules—it’s about future-proofing your role in Malaysia’s healthcare infrastructure.
Q1: How to check NPRA product search?
Answer:
You can use the NPRA Product Search via the NPRA official website. Go to the “Product Registration” section, enter the product name or registration number (MAL code), and review the official status listed in the database.
Q2: How can I verify a product’s NPRA KKM registration?
Answer:
Check the product’s MAL number (e.g., MAL12345678T) and search it on the NPRA database. A valid product will show the registration holder, manufacturer, classification, and approval status.
Q3: What is NPRA?
Answer:
NPRA stands for the National Pharmaceutical Regulatory Agency, the Malaysian authority responsible for regulating the quality, safety, and efficacy of medicines, cosmetics, supplements, and health products.
Q4: What products need a KKM check?
Answer:
Any product claiming therapeutic effects—such as medicines, supplements, traditional products, medical devices, and cosmetics—must undergo a KKM check to ensure they are registered and approved by NPRA.
Q5: What is NPRA KKM?
Answer:
NPRA KKM refers to the National Pharmaceutical Regulatory Agency under the Ministry of Health (KKM). It manages product registration, safety monitoring, and regulatory compliance for health-related products in Malaysia.
Q6: How to get KKM approval for a product?
Answer:
You must apply through the NPRA QUEST system, submit required documentation, provide product safety and quality data, pay the necessary fees, and wait for NPRA evaluation before approval is granted.
Q7: What is a KKM-approved product?
Answer:
A KKM-approved product is one that has passed NPRA’s safety, quality, and efficacy assessments and has been issued a valid MAL registration number for sale in Malaysia.
Q8: What is NPRA for?
Answer:
NPRA oversees the regulation of pharmaceuticals, cosmetics, traditional medicines, and health products to ensure public safety. It evaluates product applications, monitors adverse events, and enforces compliance standards.
Q9: How to register NPRA in Malaysia?
Answer:
Companies must create a QUEST account, prepare technical documents (COS, GMP, test reports), submit an application for product registration, and follow NPRA’s evaluation process until approval is granted.
Q10: How do I check the status of my NPRA product?
Answer:
Log in to your NPRA QUEST account and view your submission dashboard. You can check whether your product is under evaluation, queried, approved, or rejected. The public can also check via the NPRA Product Search database.
Our marketing and sales teams use their strong relationships with the channel to create demand for your product at every stage of its lifecycle.
Demand creation services we offer: