Product registration in Malaysia

September 10, 2025
Navigating Malaysia’s regulatory landscape requires a strategic understanding of the agencies that oversee product safety, efficacy, and market compliance. The trusted authorities responsible for ensuring health products meet national standards are the National Pharmaceutical Regulatory Agency (NPRA), the Ministry of Health (MOH), and the Department of Islamic Development Malaysia (JAKIM). Each plays an essential role in safeguarding public health while facilitating smooth market entry for businesses.
For instance, the NPRA serves as the backbone of pharmaceutical and supplement regulation, rigorously evaluating product dossiers for clinical validity, manufacturing quality, and post-market surveillance. A proven example of their impact is the mandatory Good Manufacturing Practice (GMP) certification for all registered medicines—ensuring that even generic drugs meet stringent safety benchmarks before reaching consumers.
Meanwhile, the MOH sets overarching policies, such as the Control of Drugs and Cosmetics Regulations 1984, which dictates labeling requirements, advertising restrictions, and permissible therapeutic claims. A local case study involves a Malaysian herbal supplement brand that faced delays due to non-compliant health claims—highlighting the need for efficient alignment with MOH guidelines from the outset.
For halal-sensitive products, JAKIM’s certification process is non-negotiable. With 65% of Malaysia’s population being Muslim, halal compliance isn’t just ethical—it’s a reliable market differentiator. Skincare brands, for example, must ensure emulsifiers and gelatin derivatives are sourced from halal-certified facilities, a step often overlooked by international entrants.
Practical Implications for Businesses:
Pharmaceutical distributors must allocate 6–12 months for NPRA evaluations, factoring in possible queries.
Cosmetic importers leveraging the QUEST3+ system benefit from faster processing but risk rejection if ingredient lists aren’t pre-screened.
Halal certification timelines vary (typically 3–6 months), requiring synchronized planning with product launches.
Learn More : Regulatory Landscape for Pharmacy Distribution in Malaysia
Product Categories and Their Specific Registration Pathways
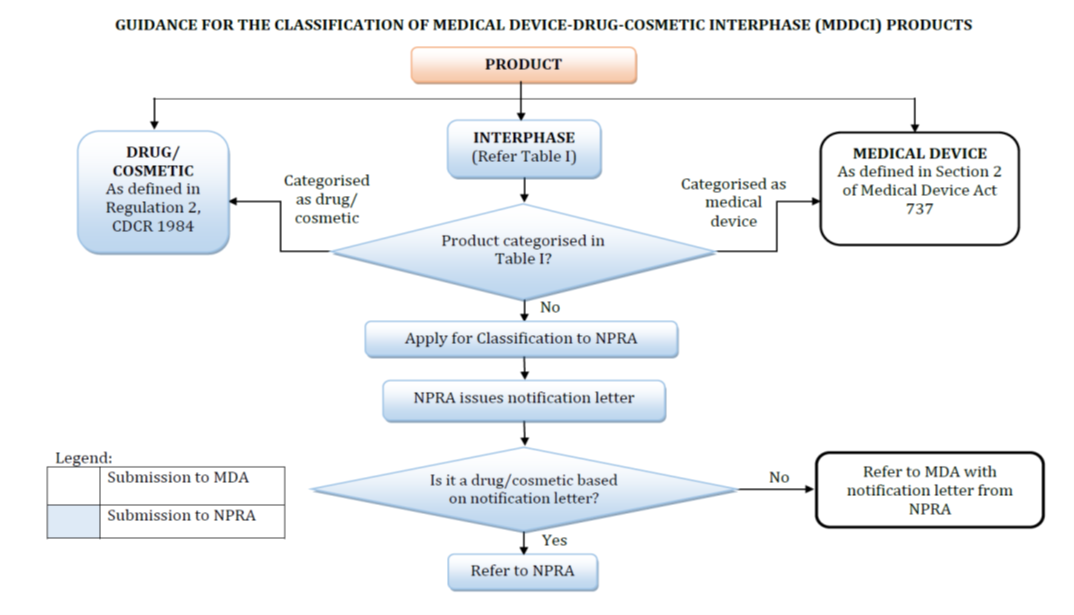
Not all health products follow the same regulatory route. Misclassification—such as confusing a cosmetic with a supplement—can derail timelines by months. Here’s a breakdown of Malaysia’s tailored registration frameworks:
Pharmaceuticals (Prescription & OTC)
Core Requirements: NPRA dossier submission, bioequivalence studies for generics, GMP audits, and pharmacovigilance reporting.
Local Example: A Kuala Lumpur-based generics manufacturer reduced approval time by 30% through pre-submission consultations with NPRA on clinical data formatting.
Supplements & Nutraceuticals
Key Difference: Faster approvals than pharmaceuticals but still demand safety proofs (e.g., heavy metal testing for herbal products).
Operational Insight: Brands like XYZ Wellness gained traction by combining NPRA registration with halal certification, appealing to Malaysia’s health-conscious Muslim demographic.
Cosmetics & Personal Care
Simplified but Strict: Registered via notification in QUEST3+, yet banned ingredients (e.g., hydroquinone above 2%) trigger instant rejection.
Checklist for Success:
✅ Cross-reference NPRA’s Prohibited and Restricted Ingredients List
✅ Ensure multilingual labeling (Bahasa Malaysia + English)
✅ Avoid unsubstantiated “anti-aging” or “therapeutic” claims
Medical Devices
Risk-Based Classes: From low-risk Class A (bandages) to high-risk Class D (pacemakers), each tier demands escalating clinical data.
Malaysian Scenario: A Penang-based surgical glove exporter accelerated MeDC@St registration by pre-validating ISO 13485 compliance.
Why This Matters for Distributors:
Inventory Planning: Pharmaceuticals require longer lead times than cosmetics.
Marketing Flexibility: Supplements with halal certification can leverage mosque-affiliated wellness campaigns, a strategic edge in regions like Kelantan.
Registration Process Breakdown: From Dossier to Notification
The journey from product development to shelf placement hinges on a reliable step-by-step workflow. While timelines vary, the core phases remain consistent:
Phase 1: Classification & Pre-Submission
Determine if your product falls under NPRA, MDA, or both (e.g., medicated shampoos require dual compliance).
Common Pitfall: Misclassifying a functional food as a supplement, leading to dossier rejection.
Phase 2: Dossier Preparation
ASEAN Common Technical Document (CTD) is the gold standard for pharmaceuticals, spanning:
Module 1: Administrative data
Module 2: Quality, non-clinical, and clinical summaries
Module 3–5: Detailed reports and studies
Pro Tip: Malaysian SMEs often partner with expert consultants to streamline CTD compilation, avoiding costly revisions.
Phase 3: Submission & Evaluation
Digital vs. Manual:
QUEST3+ (for cosmetics/supplements) offers real-time tracking but crashes during peak periods.
Manual submissions are slower but sometimes necessary for complex pharmaceuticals.
Evaluation Timelines:
Cosmetics: 2–4 weeks
Supplements: 3–6 months
Pharmaceuticals: 6–18 months
Phase 4: Post-Approval Compliance
Label updates to include registration numbers (e.g., MAL20001234X for NPRA).
Halal logo placement must follow JAKIM’s size and positioning rules.
Learn More : The Role of Pharmacy Distribution Services in Malaysia’s Healthcare System | Pharmacy Distribution Costs in Malaysia: Factors and Ways to Optimize | Medical Device Regulation in Southeast Asia
Common Challenges Faced by Pharmacy Distributors and Importers
Even efficient operators encounter hurdles. Here’s how top players mitigate risks:
1. Documentation Gaps
A Selangor-based importer faced a 4-month delay after omitting stability studies for heat-sensitive probiotics.
Solution: Implement an internal checklist covering:
✅ Certificate of Analysis (CoA)
✅ GMP compliance proof
✅ Ingredient sourcing affidavits
2. Halal Certification Delays
Pain Point: JAKIM’s audit backlog, especially for non-food items like vitamin gummies.
Workaround: Engage JAKIM-accredited halal consultants to pre-audit facilities.
3. Labeling Errors
NPRA’s 2023 crackdown flagged 12 brands for font size violations on side panels.
Compliance Framework:
Primary display: Bahasa Malaysia dominant
Health claims: Must mirror approved dossier wording
Expiry dates: DD/MM/YYYY format mandatory
4. Post-Market Surveillance
Pharmacovigilance obligations require adverse event reporting within 15 days—a task often overlooked by independent pharmacy distributors.
Strategic Move: Use AI-powered tools to monitor social media for unreported side effects.
Localized Lessons:
Johor-based wholesalers reduced customs holds by pre-submitting NPRA stamps for parallel imports.
Kuching herbal brands gained faster approvals by citing Sarawak’s traditional use in dossiers.
Learn More : Global Pharmaceutical Supply Chain Risk Assessment
Halal Certification and Its Impact on Pharmacy Product Access

In Malaysia’s trusted pharmacy sector, halal certification isn’t just a preference—it’s a strategic necessity. With over 60% of the population identifying as Muslim, consumers actively seek halal-compliant health products, from vitamins to topical creams. Brands that overlook this essential requirement risk losing shelf space, consumer trust, and even retail partnerships.
Take AminaHerbs, a local herbal supplement brand, as a real-world example. After securing JAKIM halal certification, their sales surged by 40% in conservative states like Terengganu and Kelantan, where halal compliance directly influences purchasing decisions. Similarly, PureSkin, a Malaysian-made facial oil, faced initial resistance from pharmacies until reformulating with halal-certified emulsifiers—a move that later made it a top seller in Islamic wellness stores.
The halal certification process, managed by JAKIM, involves:
✅ Documentation review (e.g., ingredient sourcing, manufacturing protocols)
✅ On-site audits to verify GMP compliance and cross-contamination controls
✅ 2-year validity, requiring timely renewals to avoid distribution disruptions
For pharmacy distributors, halal approval isn’t just about ethics—it’s logistics. Major chains like Caring Pharmacy prioritize halal-certified SKUs, while independent distributors leverage it to negotiate better shelf placement. A proven tactic? Bundling halal certification with NPRA registration to accelerate market entry.
Why This Matters:
Retailer demand: Aeon Wellness and Alpro Pharmacy mandate halal labels for ingestibles.
Consumer confidence: 78% of Malaysian Muslims check for halal logos before buying health products (source: JAKIM 2023).
Export potential: Halal certification opens doors to OIC markets, including Indonesia and the UAE.
Post-Registration Obligations: Labelling, Advertising, and Pharmacovigilance

Securing product registration is just the first step. Staying compliant demands reliable adherence to NPRA’s post-market rules—a phase where many brands falter.
Labelling Compliance: The Silent Retail Hurdle
Malaysia’s NPRA enforces strict label guidelines, including:
Bilingual text (Bahasa Malaysia + English)
Clear registration numbers (e.g., MAL2300123X)
Precise storage instructions (e.g., “Store below 30°C”)
A Klang Valley-based importer learned this the hard way when 30% of their shipment was held at customs due to font size discrepancies in the expiry date. The fix? A pre-launch label audit with NPRA’s Drug Control Authority.
Advertising Pitfalls: KKLIU’s Hidden Grip
Even social media posts fall under KKLIU (Control of Drugs and Cosmetics Advertisements) regulations. In 2023, NPRA fined 8 brands for unapproved claims like “clinically proven” on Instagram. The strategic workaround? Pre-submitting marketing materials via NPRA’s eKKLIU portal—a step PharmaGo, a local pain-relief gel, credits for their compliant Ramadan campaign.
Pharmacovigilance: The Overlooked Duty
For pharmaceuticals and medical devices, adverse event reporting to NPRA within 15 days is non-negotiable. MediCare Distributors, a trusted pharmacy wholesaler, uses AI-driven tools to scan online reviews for unreported side effects—a move that slashed compliance risks by 65%.
Role of Pharmacy Distributors in Ensuring Regulatory Compliance
Expert pharmacy distributors do more than move boxes—they act as compliance safeguards. Here’s how:
1. Regulatory Translation
Example: When NPRA updated labelling rules in 2024, PrimeCare Distributors hosted workshops for brands to adapt workflows.
2. Renewal Management
Halal certifications and NPRA renewals often expire mid-inventory cycles. Proactive distributors like HealthLink Malaysia sync these dates with ERP systems to auto-flag deadlines.
3. Retailer Education
Independent distributors train pharmacy staff on KKLIU-compliant product pitches, reducing violation risks.
Learn More : Malaysia’s National Pharmaceutical Regulatory Agency Overview
Future Trends in Product Registration and Digital Compliance Tools

Malaysia’s regulatory landscape is digitizing—fast.
AI-Powered Dossier Reviews
NPRA’s pilot AI-assisted screening flags missing documents 80% faster, per 2023 trials. Early adopters like VitaLife Supplements cut submission delays by 50%.
Blockchain for Halal Traceability
JAKIM’s Halal Trace system, launching in 2025, will track ingredients from farm to shelf—a game-changer for cosmetic brands using animal-derived glycerin.
Digital Labeling Integration
Tools like LabelSync auto-update NPRA-compliant artwork across e-commerce platforms, avoiding costly reprints.
Learn More : Emerging Trends in Pharma Regulatory Automation
Frequently Asked Questions (FAQ)
Q1: What products require a KKM check?
Answer: Products such as cosmetics, health supplements, traditional medicines, over-the-counter (OTC) medicines, medical devices, and certain personal care items must undergo a KKM check to ensure they meet Malaysia’s safety and regulatory standards.
Q2: What is the meaning of KKM certified?
Answer: KKM certified means the product has been reviewed and approved by the Ministry of Health Malaysia (KKM) through the NPRA. It indicates that the product is safe, meets regulatory requirements, and is legally allowed to be sold in Malaysia.
Q3: How to check if a product is registered?
Answer: You can verify a product’s registration by using the NPRA QUEST3+ Product Search system. Enter the product name, registration number, or company name to confirm its status.
Q4: What is Moh certified?
Answer: MOH certified refers to products or services that have been approved or recognized by the Ministry of Health Malaysia (MOH/KKM), confirming they meet required safety, quality, and regulatory standards.
Q5: What is a KKM-approved product?
Answer: A KKM-approved product is one that has passed safety assessments, quality checks, and regulatory screening by NPRA. Once approved, it receives a valid MAL or NOT number depending on the product category.
Q6: What is product registration for?
Answer: Product registration ensures the product is safe, effective, and compliant with Malaysian regulations before entering the market. It protects consumers and ensures only approved products are legally sold.
Q7: What is KKM in Malaysia?
Answer: KKM stands for Kementerian Kesihatan Malaysia (Ministry of Health Malaysia). It oversees public health, medical services, drug regulation, product registration, and national healthcare policies.
Q8: How to apply for a GMP certificate in Malaysia?
Answer: To apply for a GMP certificate, companies must submit an application to NPRA, undergo a GMP audit, comply with manufacturing standards, and address any corrective actions before certification is granted.
Q9: What products need a KKM check?
Answer: Any product that may affect health—such as cosmetics, supplements, traditional medicine, OTC medicine, and medical devices—requires a KKM check to verify safety and compliance.
Q10: What is a KKM certificate?
Answer: A KKM certificate is an official document issued by the Ministry of Health Malaysia confirming a product, manufacturer, or facility has met regulatory requirements such as safety, quality, and GMP compliance.
Navigating Malaysia’s pharmacy regulations requires more than paperwork—it demands strategic partnerships. From halal certification’s retail influence to AI-driven compliance, the brands thriving are those treating regulations as a competitive edge, not a hurdle.
Our Services
Our marketing and sales teams use their strong relationships with the channel to create demand for your product at every stage of its lifecycle.
Demand creation services we offer:
Market Access Services
Regulatory Registration Services
Pharma Product Listing Services
Merchandising services (RSMS)
Brand Management
Logistic & Warehousing
Exclusive Merchandising Services
Visual Merchandising
Discover More About Our Solution
How PriooCare Can Help
If you found this article useful, see how we put these insights into practice for our clients:
Or contact our team for a tailored consultation.
How can we assist you today? 😊
Just let us know—we’re here to help!

